Optimising Fluid Therapy in the Critically Ill
Introduction to the 7D conceptual framework
Watch PeerVoice presentation
Learning Objectives
-
Describe the current recommendations for fluid management and how they apply in the real world
-
Recognise the variations in crystalloid and colloid fluids and their appropriate use
-
Translate the latest evidence for fluid replacement and its potential impact on clinical strategies
Manu Malbrain, MD, PhD: co-founder and President of the International Fluid Academy, and will give a talk on everything you need to know about fluid therapy in the critically ill.
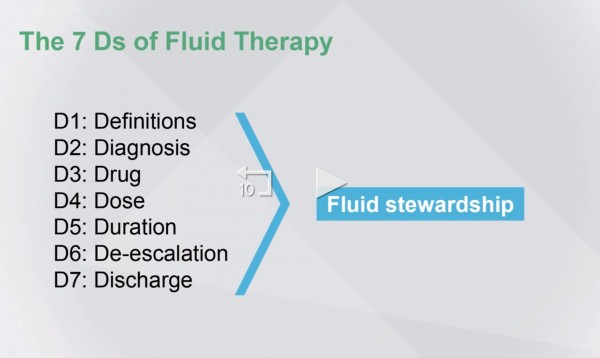
A conceptual framework will be used of seven Ds: definitions, diagnosis, drug, dose, duration of treatment, de-escalation of fluid therapy, and when the patient is [leaving the ICU or] going back home (discharge).
When we multiply, then we get D to the seventh power, or fluid stewardship. And, this is the topic of this presentation.

Abbreviation(s): EAFM: early adequate fluid management; LCFM: late conservative fluid management; LGFR: late goal-directed fluid removal; ICU: intensive care unit; SSCG: Surviving Sepsis Campaign Guideline. Reference(s): Malbrain MNLG et al. Ann Intensive Care. 2018;8:66. doi: 10.1186/s13613-018-0402-x.
D1 is all about definitions, and without going into detail it must be mentioned that fluid overload is defined as a cutoff value of 10% fluid accumulation. And, this is associated with worse outcomes.
We also have dynamic phases of fluid management, and will also talk about late goal-directed fluid removal, which is active fluid removal by means of diuretics or renal replacement therapy with net ultrafiltration. And, this is what we refer to as de-resuscitation.
Reference(s): 1. Malbrain MNLG et al. Ann Intensive Care. 2018;8:66. doi: 10.1186/s13613-018-0402-x.
2. Vincent JL, Pinsky MR. Critical Care. 2018;22:214.
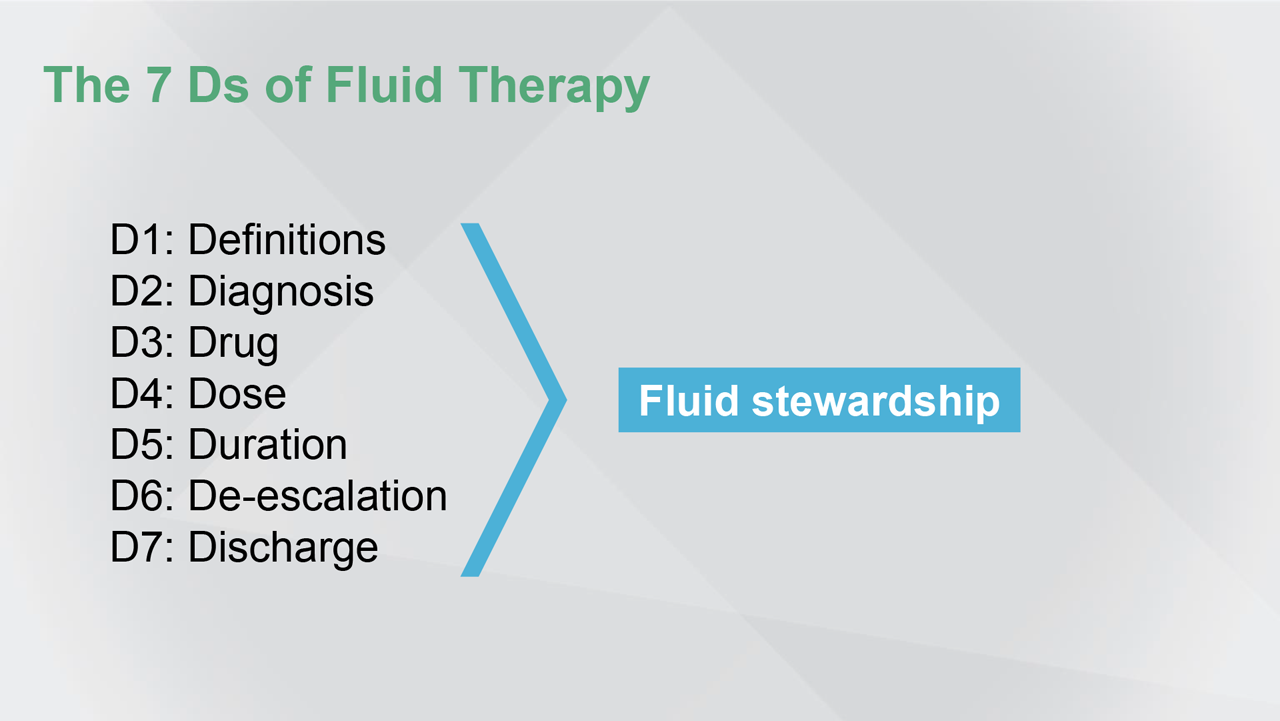
Fluid overload usually leads to hypervolemia, hyperhydration, and peripheral oedema. But, while hypervolemia is usually associated with oedema, the presence of oedema does not mean that the patient is hypervolemic. So, maybe we should avoid the term ‘fluid overload.’
Abbreviation(s): IAP: intra-arterial pressure.
Reference(s): Bellamy MC. Br J Anaesth. 2006;97:755-757.
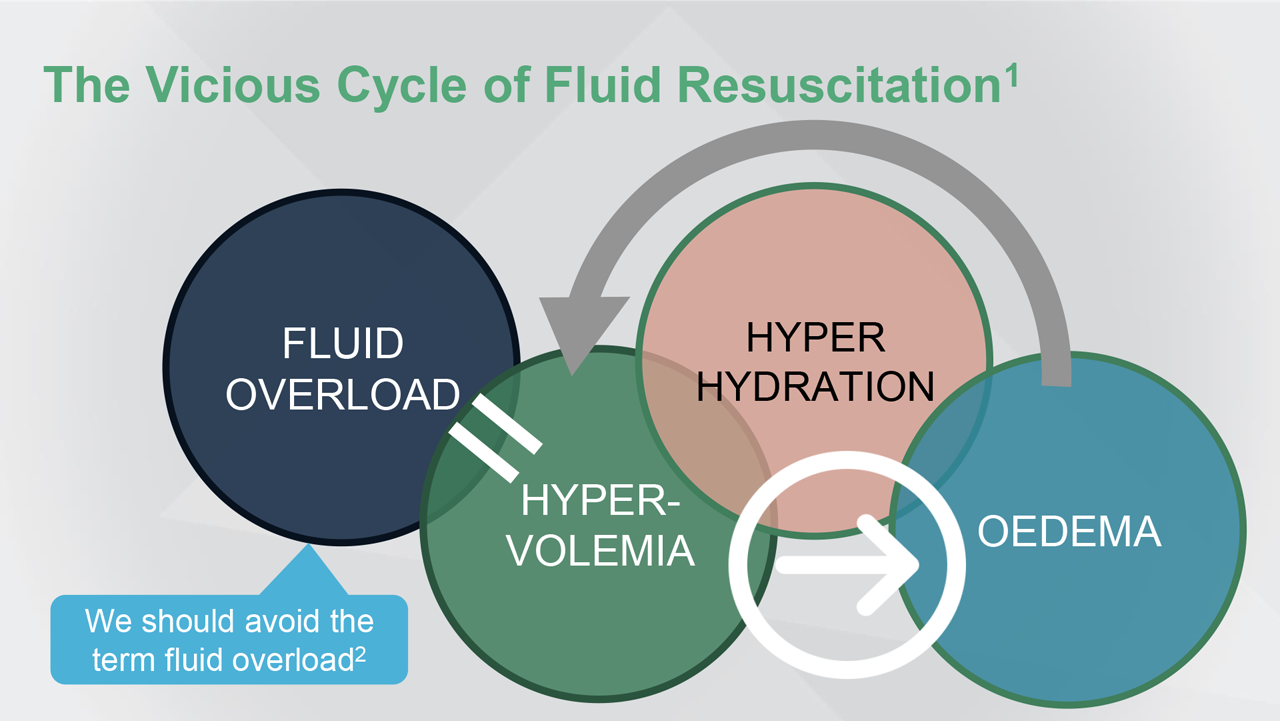
D2 is diagnosis. Hypovolemia is bad, but hypervolemia may even be worse. So, it’s finding the balance between hypovolemia, where convective problems may arise and oxygen cannot get to the tissue, or hypervolemia, where there will be diffusion problems and oxygen can’t get to the tissues either. But, what is normovolemia?
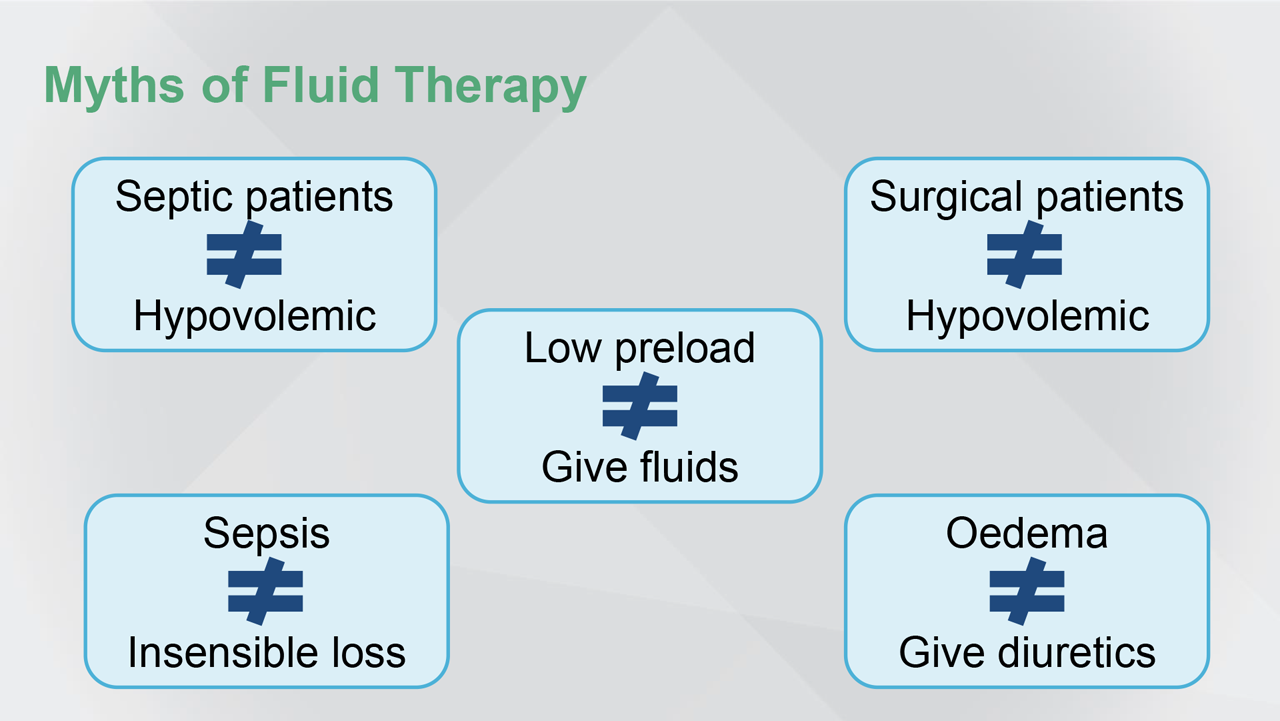
The fluid status matters, and there are a lot of myths surrounding fluid therapy. Septic patients are not always hypovolemic. Surgical patients are not always hypovolemic. Sepsis does not mean that there are huge insensible losses. Peripheral oedema does not mean that we need to give diuretics. A low preload does not mean that we need to give fluids in all cases.

This brings me to D3, which is all about the drug. First of all, we must avoid doing harm to the patients and ask ourselves four questions—the first one being when to start fluids because, after all, if the patient doesn’t need fluids, why
give them unnecessarily? The second question is when to stop IV fluids. Third: in those patients that do not transgress spontaneously from the ebb to flow phase, we need to start active fluid removal or de-resuscitation. And, the fourth question to avoid hypovolemia and hypoperfusion is when to stop fluid removal.
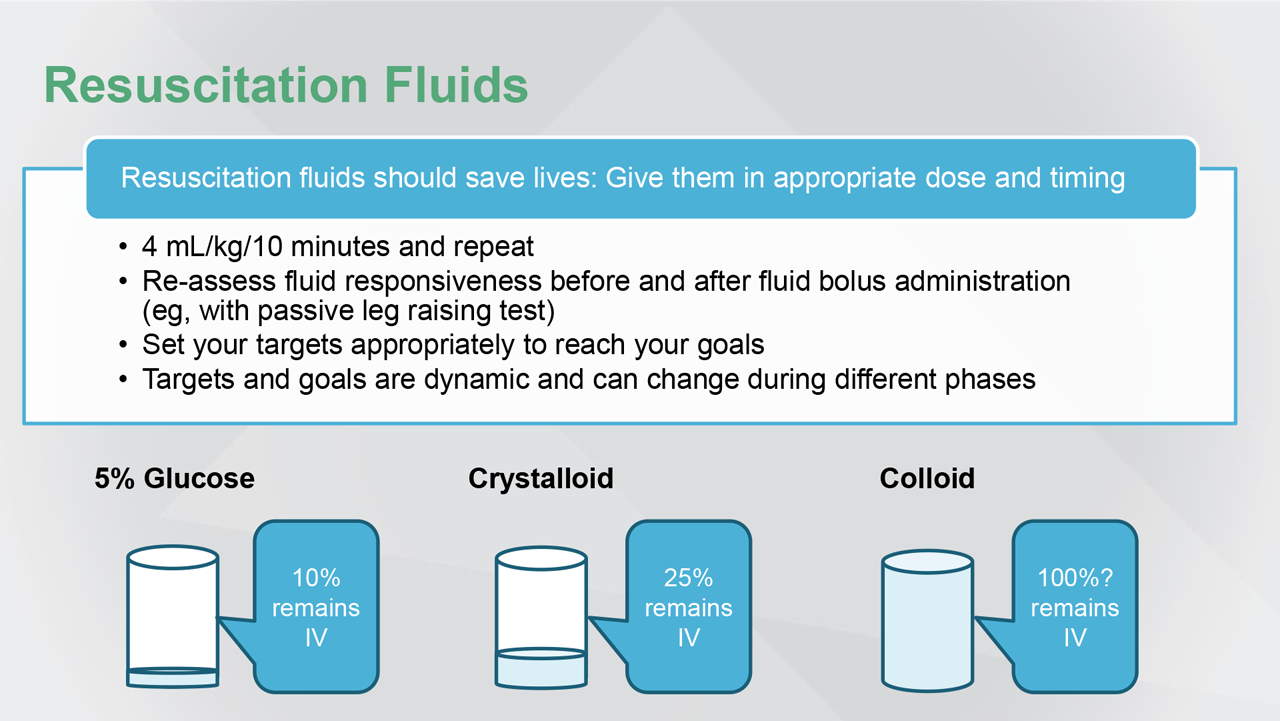
There are only four indications to give fluids—[the] first
one being for resuscitation. And, those fluids should save lives. The bottom line may be that a hypotonic solution like glucose 5%, when administering it for resuscitation, only 10% will remain intravenously after 1 hour. [Therefore hypotonic solutions should not be used for resuscitation; they can be used in case of cellular dehydration.] For crystalloids, either balanced or normal saline, about 25% to 30% will remain IV after 1 hour. And, we assume that colloids will remain 100% IV after 1 hour.

So, putting all the evidence together, when it comes to fluids and sepsis in critical care, it’s probably not a good idea to give starches, gelatins, saline in large amounts, or hypotonic solutions like glucose 5%. Balanced crystalloids are a good first choice. And, of course, blood should be given, taking into account the shelf life when needed. Albumin 20% may have a role in de-resuscitation. After all, the fluid strategy is more important than the fluid itself.
Reference(s): Van Regenmortel N et al. Intensive Care Med. 2018;44:409-417.
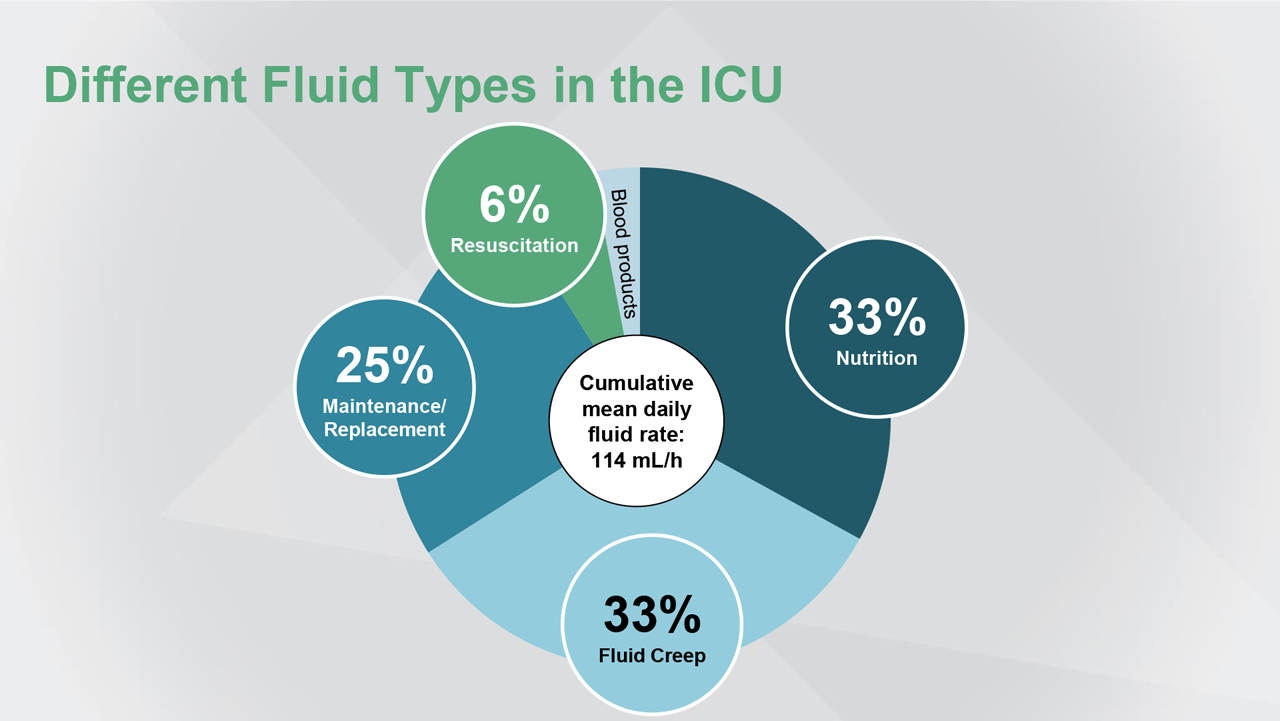
While resuscitation fluids received the most attention, the largest part of fluids that we give to our critically ill patients is maintenance fluids and other fluids.
In this study by Van Regenmortel of almost 15,000 patients, it was found that only 6% of fluids administered were for resuscitation. Twenty-five percent were maintenance, 33% nutrition, and 33% was defined as fluid creep—fluids that seep into the patient for administration of drugs, pain killers, antibiotics, and so forth.

Maintenance fluids should cover the daily needs of water, glucose, sodium, [and] potassium. Water need is about 1 mL/kg/hour. Glucose need is about 1.0 to 1.5 g/kg/day, and sodium/potassium needs are [around] 1 mmol/kg/day.
Abbreviation(s): CI: confidence interval.
Reference(s): Van Regenmortel N et al. Intensive Care Med. 2019;45:1422-1432.
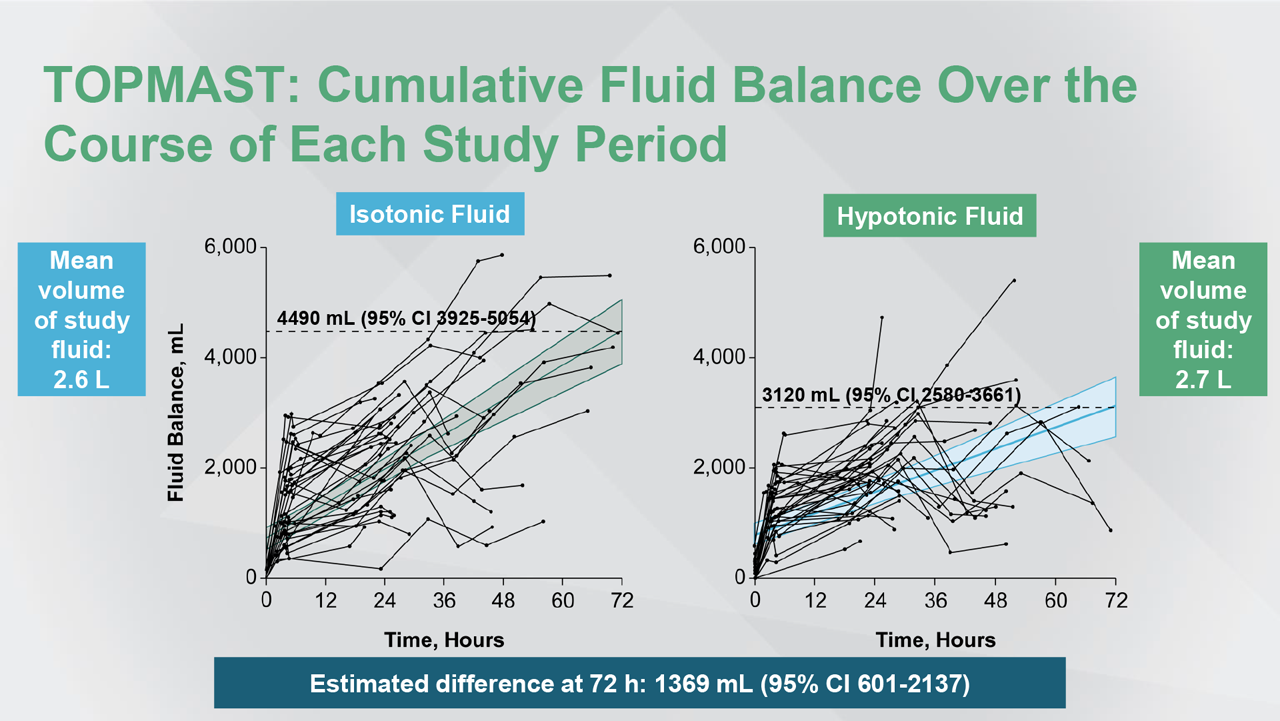
We studied the effects of hypotonic versus isotonic maintenance solutions in critically ill patients undergoing thoracic surgery. And, we found after 3 days that hypotonic maintenance solutions resulted in a less positive cumulative fluid balance of about 1.5 litres [less positive over the period of 3 days].

Abbreviation(s): GI: gastrointestinal.
The third indication is replacement fluids, and those should mimic the fluid that is lost. And, gastrointestinal losses may be the only indication left for what I would call abnormal saline, [considered abnormal because we don’t have plasma sodium nor chloride levels of 154 mmol/L]. Nutrition fluids should cover the caloric needs. Those are the four indications.

So, referring to the four Ds in analogy to antibiotic stewardship—drug, dose, duration, de-escalation—we can also translate this to fluids as drugs. The dose is most important, as the dose will determine whenever something becomes poisonous.
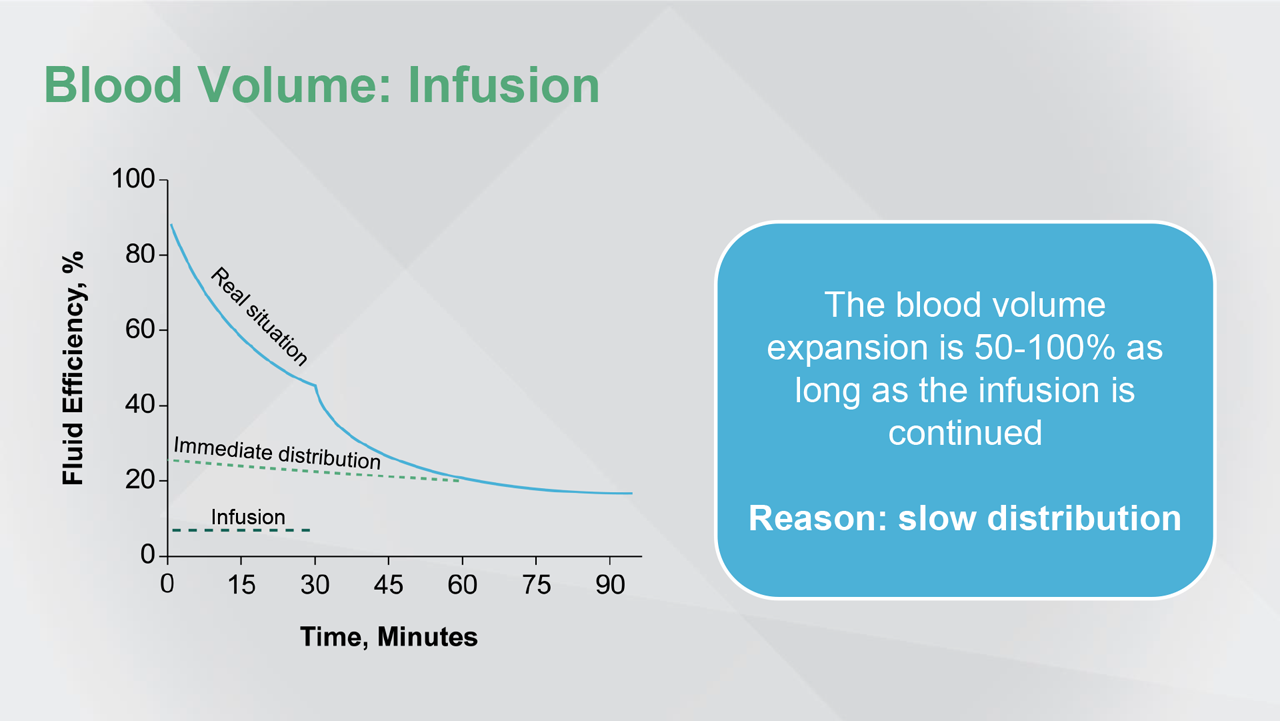
Reference(s): Hahn RG. Acta Anaesthesiol Scand. 2013;57:16-28.
The dose refers to pharmacokinetics and pharmacodynamics. So, as long as IV fluids are infused, there will be blood volume expansion. And, the reason for this is the slow distribution.
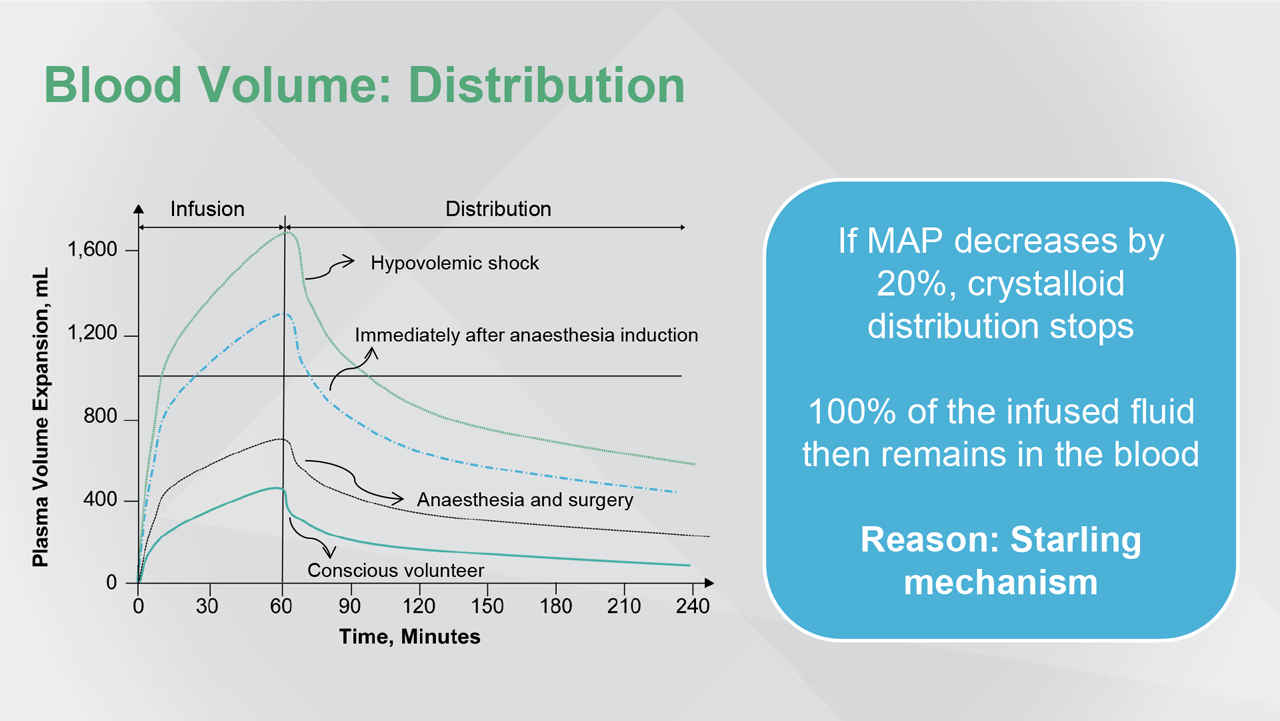
Abbreviation(s): MAP: mean arterial pressure.
Reference(s): Hahn RG. Acta Anaesthesiol Scand. 2013;57:16-28.
Distribution of IV fluids decreases by 20% to 100% if blood pressure drops as little as 20%. [As a result, crystalloids and colloids will have the same volume expansion effect.] The reason for this is Starling mechanisms.
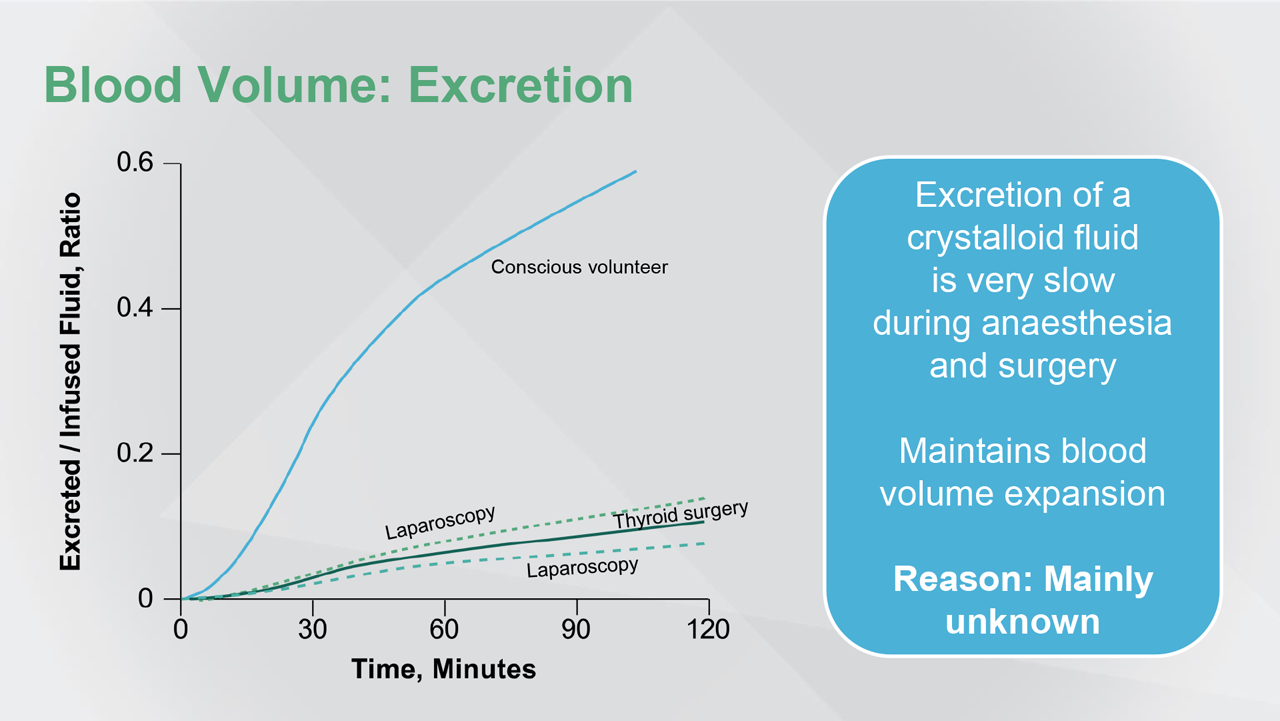
Reference(s): Vincent JL (ed). Annual Update in Intensive Care and Emergency Medicine 2015. Switzerland: Springer International Publishing; 2015.
Hahn RG. Clinical Implications From Dynamic Modeling of Crystalloid Fluids; pp 339-348.
And, excretion of a crystalloid fluid is very slow during anaesthesia and surgery and shock states. So, this could mean that crystalloid fluids may have the same blood volume expansion effect as colloids in critically ill patients or in the perioperative phase.

D5 is duration. So, we should stop fluids when they are no longer needed, and the patient can have an oral fluid intake.
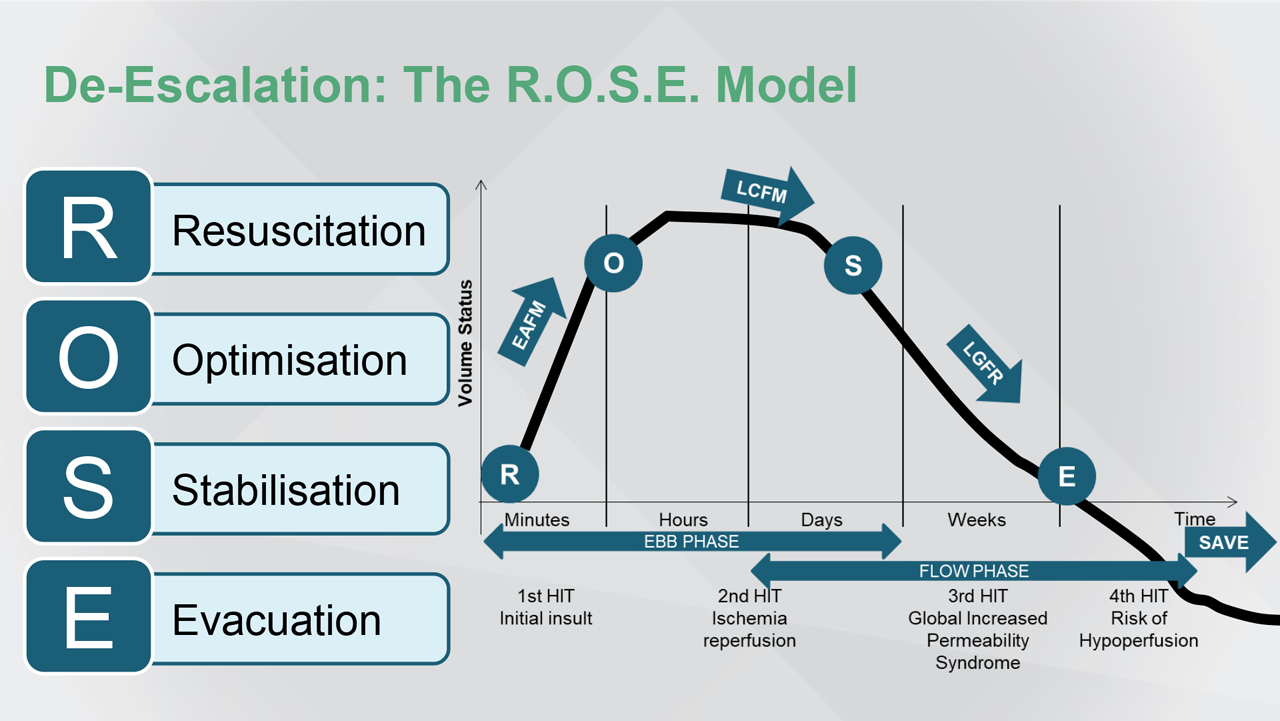
Reference(s): Malbrain MLNG et al. Ann Intensive Care. 2020;10:64.
Monteiro JN, Goraksha SU. J Neuroanaesthesiol Crit Care. 2017;4:10-16.
D6 is de-escalation, which brings me to the R.O.S.E. conceptual model and the four phases of fluid resuscitation, optimisation [and] organ support, stabilisation, and evacuation. So, whenever there is a first hit—whether it is trauma, sepsis, burns, pancreatitis—fluids must be given to resuscitate the patient and to save lives. And, fluid balance must be positive.
When the patient stabilises, fluids should be kept to maintain homeostasis and to support the organs. And, this is referred to as the second hit, which is about ischaemia reperfusion. The third hit is global increased permeability syndrome,
with fluids accumulating where active fluid removal may be needed. The fourth hit must be avoided, as avoiding hypoperfusion. Hence, the ROSE conceptual model.
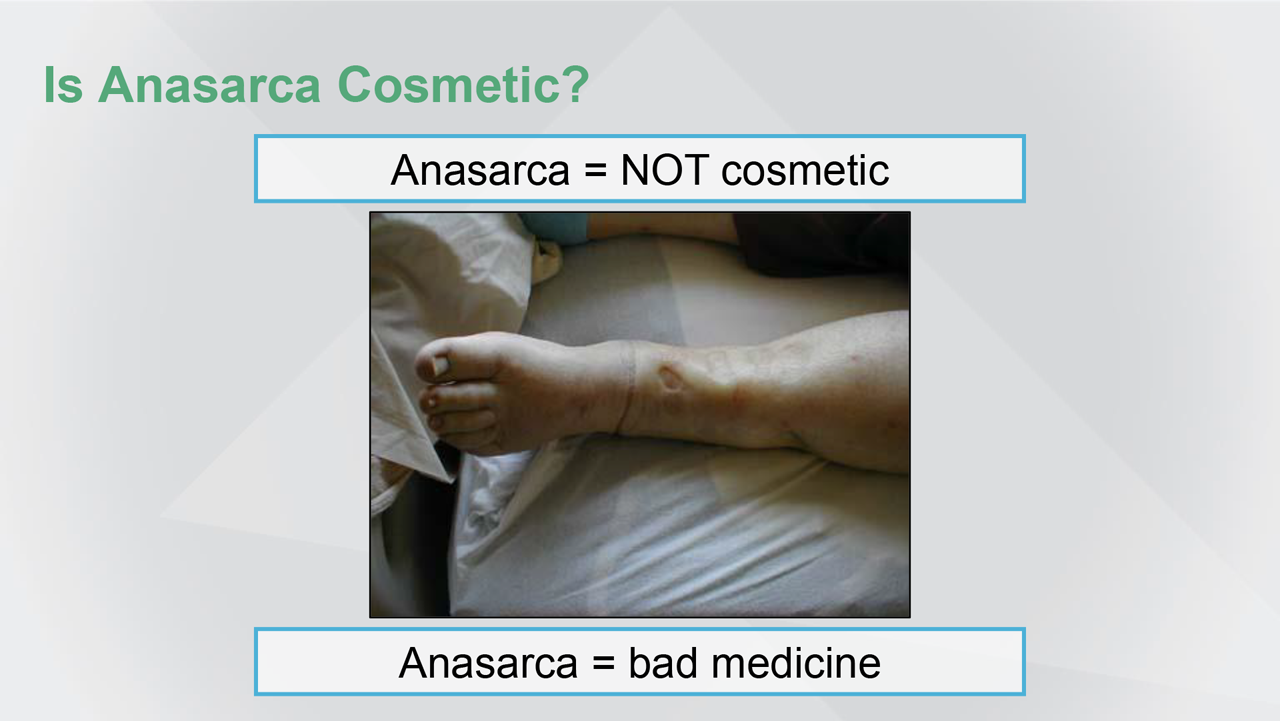
Reference(s): Photo courtesy of Manu Malbrain, MD, PhD.
The goal is to avoid this global increased permeability syndrome because peripheral oedema is not just cosmetic. It is bad medicine. Why is that?

Abbreviation(s): ACS: abdominal compartment syndrome; APP: abdominal perfusion pressure; CARS: cardio-abdominal- renal syndrome; CO: cardiac output; CPP: cerebral perfusion pressure; CS: compartment syndrome; CVP: central venous pressure; GEDVI: global end-diastolic volume index; GEF: global ejection fraction; GFR; glomerular filtration rate; IAH: intra-abdominal hypertension; IAP: intra-abdominal pressure, ICG-PDR: indocyanine green plasma disappearance rate,
ICH: intracranial hypertension, ICP: intracranial pressure;
ICS: intracranial compartment syndrome; IOP: intra-ocular pressure; OCS: ocular compartment syndrome, PAOP: pulmonary artery occlusion pressure; pHi: gastric tonometry; RVR: renal vascular resistance; SV: stroke volume. Reference(s): Malbrain MLNG et al. Anaesthesiol Intensive Ther. 2014;46:361-380.
Prowle JR et al. Nat Rev Nephrol. 2010;6:107-115.
Because fluid overload has an independent impact on outcome in patients with sepsis. And, restrictive fluid therapy results in a better prognosis. The reason for this is that fluid overload at the outside (generalised peripheral oedema, or anasarca) mirrors fluid overload at each and every organ. And, this will lead to multiple organ system failure.

Reference(s): Malbrain M et al. ICU Management and Practice. 2018;183:158-162.
This brings me to D7, discharge. And, I think it’s time for fluid stewardship, which is a series of coordinated interventions introduced to select the optimal fluid, dose, duration of therapy that results in the best clinical outcome, prevention of adverse events, and cost reduction.
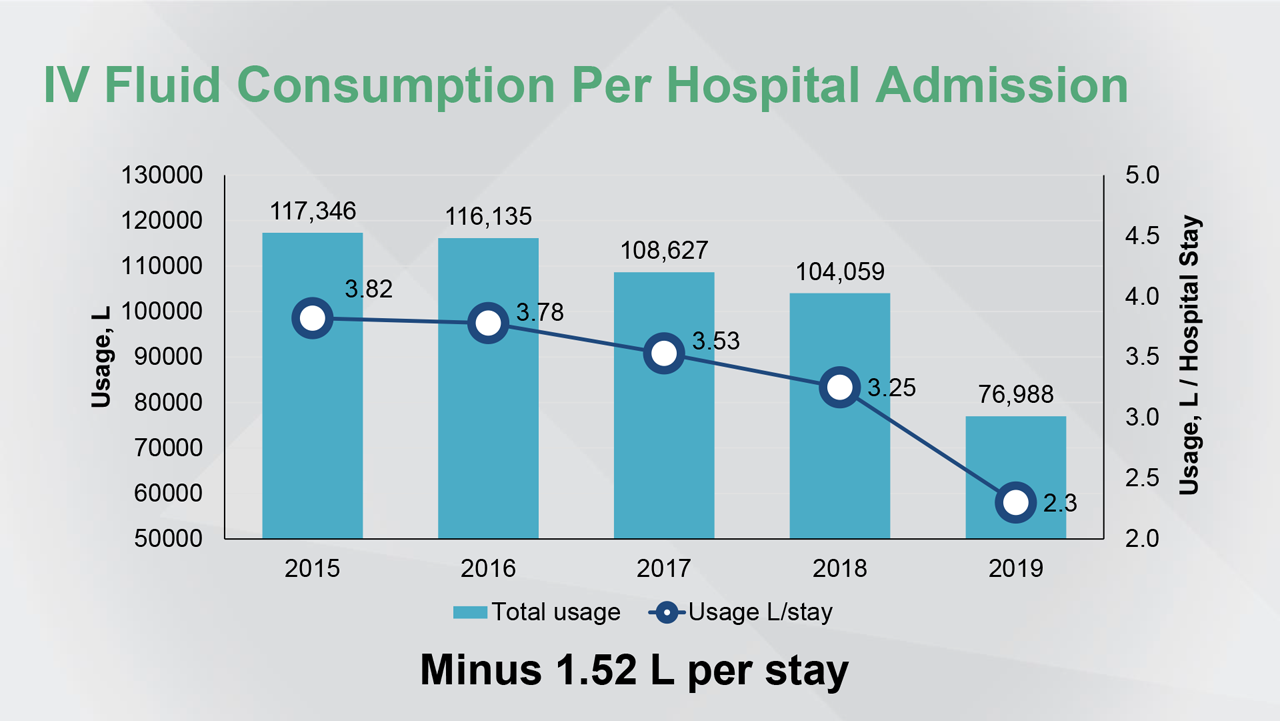
Reference(s): Personal correspondence with Manu Malbrain, MD, PhD.
So, what happened in Brussels [at University Hospital Brussels]? Over the last 5 years, we saw a significant decrease in the amount of fluids given per stay of 1.5 litre, which is about 0.2 litres less per day.
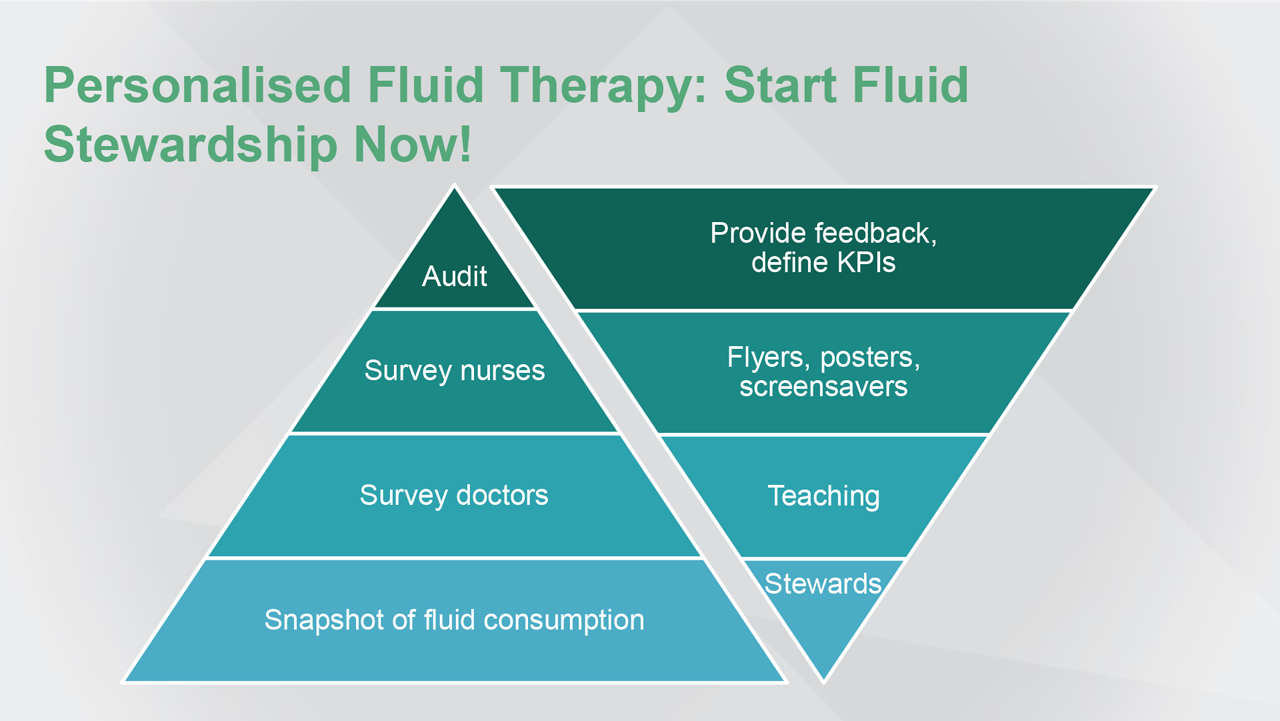
Abbreviation(s): KPI: key performance indicator.
So, I hope I convinced you that IV fluids are not just innocent bags of water, but they are drugs. And, personalised fluid therapy is needed in critically ill patients. For this, you need a fluid team with fluid stewards—doctors and nurses—on the floor.
[Therefore], I think it’s time to act now and to start fluid stewardship in your ICU. This brings me to the end of my presentation. Thanks for joining.
This PeerVoice independent medical education activity has been endorsed by the International Fluid Academy.
Content developed in concert with the faculty. Release Date: 1 December 2020